We Enable
We enable our customers to interpret the processes they undertake by bridging the physical environment with the digital world, so that they can monitor and manage their business processes more effectively.

We Focus
We typically focus on extreme environments or processes, which are regulated, and Kustodian provide a technology layer, known as an execution system, to automate tracking, tracing and process data collection.
Our Goal
Our goal is to provide our customers with safer processes, more productive people and higher quality products. By doing this we aim to create more satisfied customer’s customers.

What We Do
Technology is changing the way we live and the way we work. We see it in our daily lives as the internet becomes more pervasive. By 2020 it’s estimated over 50 billion devices will be connected to the internet; a recent Gartner estimate forecasts 30% year on year growth of connected devices. Either way the IoT will revolutionise the way we work as well as raise productivity to levels not seen since the Industrial Revolution. This means new services can be created driving ever greater collaboration and productivity. In the case of Kustodian in also means greater safety, higher productivity and lower operating costs for users of our technology.
Our mission
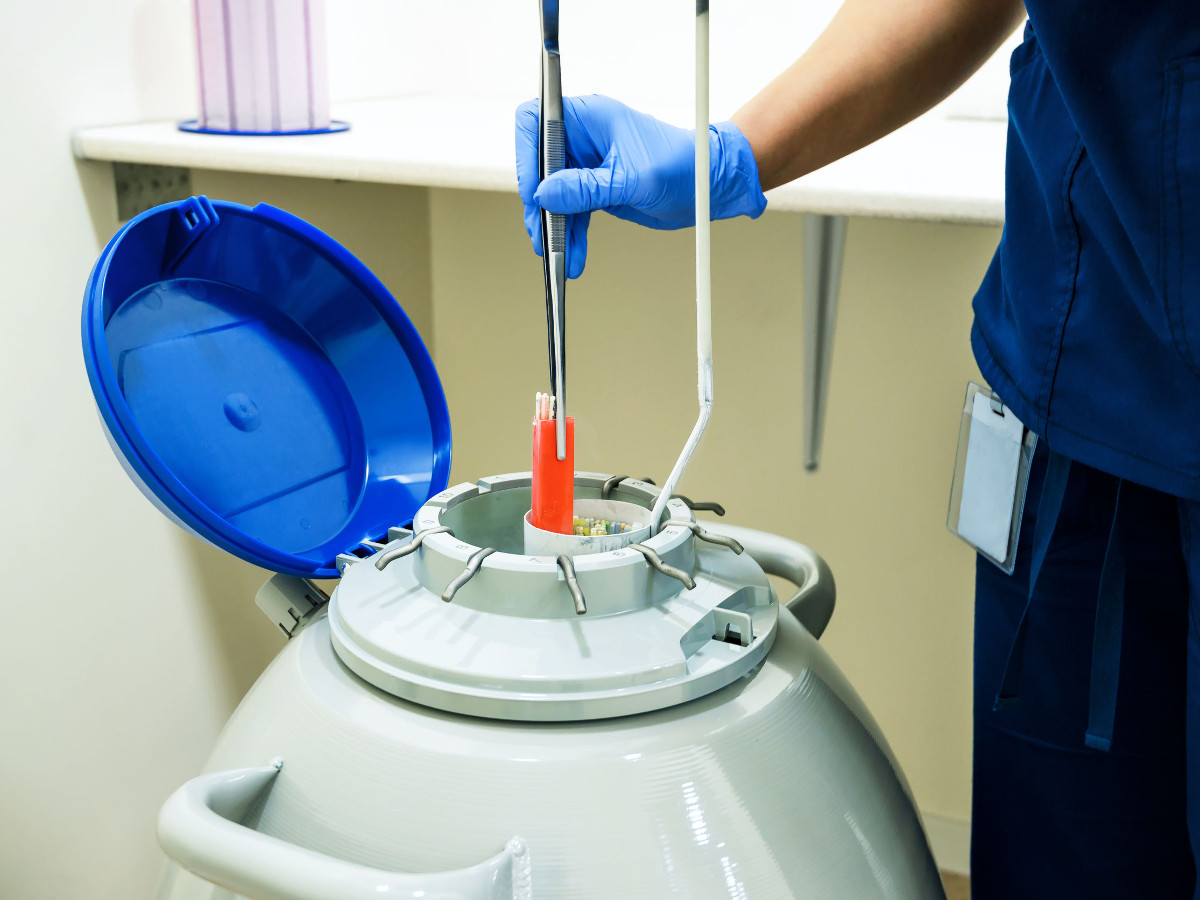
Today we are focused on processes in the field of ART – Assisted Reproductive Techniques – but our mission is to provide an asset tracking and execution system that supports any process operating in an extreme environment. Deep sea exploration, space travel and human tissue processing and tracking at -1960C are examples of extreme environments in which we offer capability.
RFID Technology
Kustodian’s patented RFID technology is built on the Internet of Things. Its modern architecture offers the exciting opportunity to revolutionise the way fertility treatments are executed and managed. The Internet of Things has already enabled AirBnB to become the largest hotel chain in the world without owning a single hotel; Uber are the largest taxi service in the world – they don’t own a single taxi. We see disparate systems in clinics, coming together so that clinicians interact with one interface to a system that gathers data about the process, the samples and the people. We envisage higher productivity, greater transparency in treatments and greater levels of patient [and staff] safety taken to new global standards.
Why Choose Kustodian
-
Kustodian Platform
The Kustodian platform provides instant feedback on whether a process has been followed correctly, when and where samples are stored and who they belong to. Finding a sample has never been easier. Ask Kustodian, and its unique Dewar based messaging hub will tell you not only in which Dewar the sample is stored but where in the Dewar the sample is located. If the sample is moved, its location is updated automatically.
-
Dewar Management
Kustodian tells you how full a Dewar is and where there is space for more samples, enabling embryologists to use Dewars in a safer and much more efficient way than is currently possible.
-
RFID Technology
Kustodian uses RFID as a labelling technology, operating in both the vapour phase and liquid phase, a global first for this technology. Field testing and extensive research over many years shows the Kustodian platform to be safe and reliable.
-
Our beliefs
We believe in the power of good design. Such an unbelievable amount of thought goes into every one of our systems because we want them to outperform your expectations.
Our way of thinking always starts with ‘people first’. We know that you have the ultimate gift to give to families. However, treatments you perform are manually managed and therefore potentially prone to error. You may not be having a good day. Your patients may not have provided good quality samples but that’s all you have to work with.
So, when we designed Kustodian, we aimed to include features and functions that make your life of making life run smoothly, with more secure processes and consistently of the highest quality.
That’s why, when we set out to design a Kustodian system for you, we always make time to imagine a day in the life of one of your clinics and ask ourselves “is there any other way to do it?”, “what else could it do?” We want it to be better than you ever imagined.
Good design isn’t solely about how perfect something looks. It runs much deeper than that. It’s about how intuitively something behaves, understanding what you need and being there, waiting, as and when you need it. It’s about how that concept then comes together with the whole process completely under control.
That’s the truth of good design - take a look at what we can do here: here.
Kustodian? ...of course we are!
-
Example Dewar
-
Assignment of Material
This video shows how the core components of the Kustodian system work and follows the initial process of assigning material to the care of Kustodian. Once this stage is complete, the sample will have a globally unique identity, based on the international GS1 labelling standards and against which all process information is recorded. This is the anchor to our world class chain of custody capability.
-
Process Execution Management
Kustodian is a process execution system, providing technology to support the complex process of tracking compliance to your standard operating procedures. This video shows you how a process (in this case, sperm preparation) is monitored, visualised and reported. By the time the sample is placed in the Dewar, Kustodian has collected all relevant information to support compliance needs and your business dependencies – consent, billing, patient records and more.
-
Auditing tissue in liquid nitrogen
Auditing the content of a Dewar is an arduous and hazardous activity. We’ve not come across any clinic that looks forward to performing the audit but without exception, every clinic we speak to recognises the importance of an audit and welcomes the innovation Kustodian brings to this critical activity. Kustodian performs an audit every time the Dewar cap is removed and replaced by reading sample tags that are immersed in liquid nitrogen. It checks to see if the outcome is as expected and reports any anomalies. The audit takes a few seconds to complete but that’s slightly quicker than any other method in use today!
-
How to check the content of a Dewar in 10s
This video demonstrates how Kustodian monitors for incorrect processing of material, with graphics depicting the content of the Dewar and sample status. Users can drill down to a specific asset (straw, vial etc) to gather itemised data about the patient’s asset.
-
Management Reports
Once you have all your samples stored, it is of course, important to know where each sample is located. Traditionally this information is held on paper records, in a spreadsheet, or in the Laboratory Information System. These are, of course, separate systems creating separate records that have to be checked, updated and maintained.
We have consolidated reporting so that all relevant information is available on one system, Kustodian. We start with the basic report – where are the samples and from this foundation, data is shared with other systems e.g. LIMS will need to know about process outcomes, Quality Control will need to know when consents expire or samples are missing; Finance will need information to support a storage billing cycle; Purchasing will need to know when consumable inventory is getting close to re-order points. All this data is held in Kustodian to support easy compliance reporting and effective business operations.
Our Benefits
-
Managing the Statutory Audit
The HFEA statutory inventory audits is actually performed by Kustodian every time you put away or remove a sample from the Dewar. Kustodian monitors this activity and adjusts inventory records accordingly using its unique Dewar based messaging hub. Kustodian support for the statutory audit is unprecedented and addresses the concerns around the accuracy and safety of this periodic, tedious yet critical process.
-
Track and Trace – total accessibility
Kustodian brings Big Data analysis to the Clinic, making patient information instantly available quickly and easily to those authorised to access the information, including patients. You are no longer reliant upon team members writing in note books. Kustodian is a highly productive and intuitive tool, using “smartphoenesque” applications to record activities associated with preparation, filling, tracking and reporting on all samples processed in your clinics.
-
Inventory Management, GS1 and Patient Safety
Kustodian uses the international standard, GS1, to identify and track samples. GS1 makes every consumable and every sample in every consumable globally unique and is a standard adopted by NHS Procurement. Kustodian provides traceability to a highly granular level – whose sample, who processed it, where the sample is, what container it’s in, who created the sample, who made the consumable and from what batch the consumable is derived. Traceability and hence patient safety is taken to a new global standard with Kustodian.
-
KRIS™ – Kustodian Konsumable Replenishment Service
We mentioned new services. Since Kustodian can track consumables throughout their life cycle, KRIS will track and optimise your consumable inventory and replenish automatically to suit your demand.
Meet Our Team
Kustodian has a strong management team who have significant experience of the market and creative ability. They have all delivered results in high quality, high visibility markets.
The directors of Kustodian bring a strong and diverse range of skills and experience. Their individual achievements throughout their careers provide evidence of self-motivation and capabilities.

Alex
Chief Executive Officer
alex.parker@kustodian.co.uk
+44 7967 581 605

Karin
Chief Clinical Embryology Officer
karin.dawson-smith@kustodian.co.uk
+44 7702 800 240

Giles
Head of Research and Development
giles.palmer@kustodian.co.uk
+44 7471 357 017

Peter
Chief Product Officer
peter.parker@kustodian.co.uk
+44 7973 285 736